PETG bottles triple-packed - GMP-compliant for cleanroom & aseptic production
Triple bagged PETG media bottles 1 litre - sterile, triple packed, for GMP class A/B/C cleanrooms and isolators
The innoME Triple Bagged PETG bottles were specially developed for use in GMP-regulated cleanrooms, aseptic production areas and isolators developed.
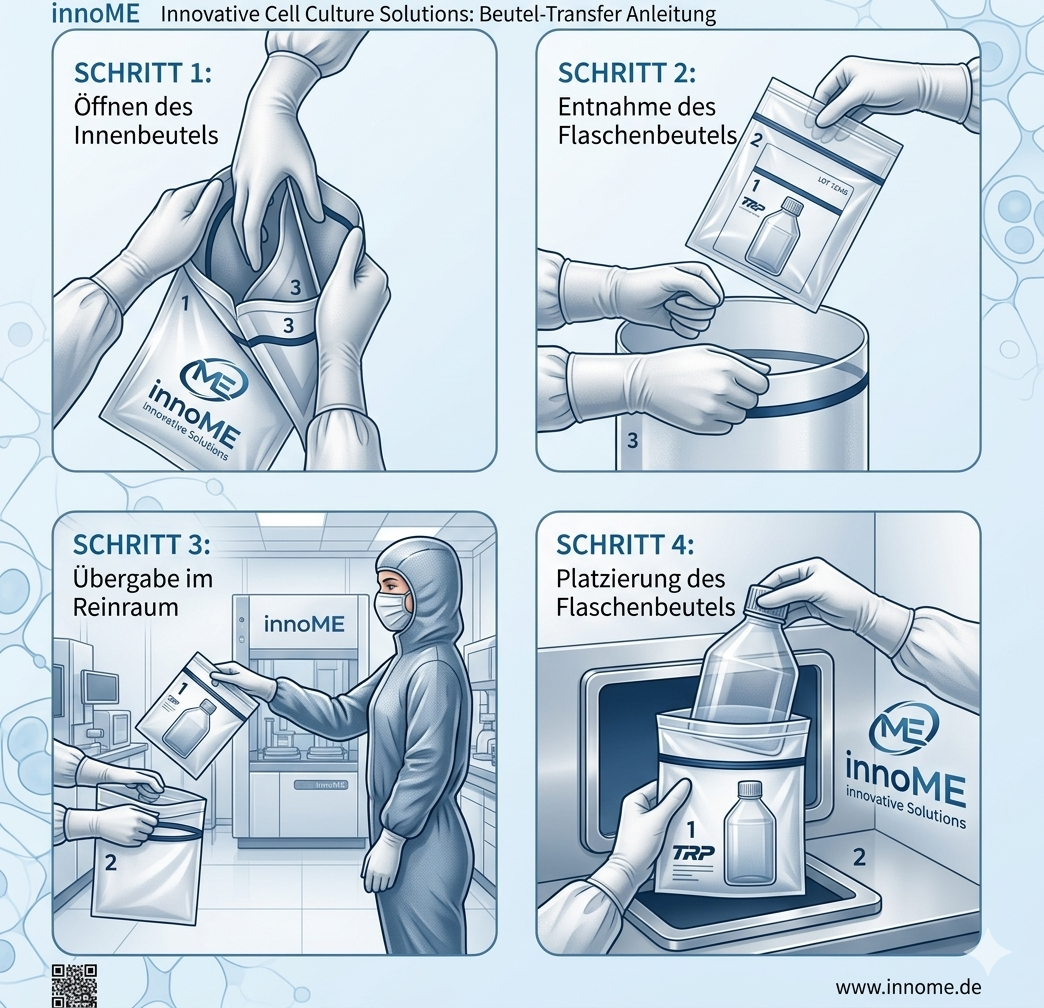
Each bottle is Triple-packed in staggered tear-off bags - Each bag is numbered (3=outside, 2=centre, 1=inside) for seamless status display when entering the cleanroom. No wiping with alcohol required, no risk of contamination.
Whether for Biopharmaceutical production, vaccine production, CGT therapies or aseptic filling - Our triple-packed PETG bottles fulfil the strictest GMP requirements for Cleanroom classes A, B, C and D as well as isolators.
Product Information
3-fold packaging (triple bagged)
Each bottle is packed in three staggered, larger tear-off bags. Each bag is numbered: 3 = outer bag (warehouse), 2 = centre bag (airlock), 1 = innermost bag (cleanroom). The size grading prevents the inner bag from being damaged when the outer bag is opened. No wiping with alcohol required.
PETG - ideal cleanroom material
PETG (polyethylene terephthalate glycol) offers high chemical resistance, excellent transparency for visual inspection and anti-leakage properties. Temperature resistant down to -70°C, sterilised by gamma irradiation (SAL 10-⁶), DNase-free, RNase-free, pyrogen-free.
GMP conformity
Fully compliant with EU GMP Annex 1 (aseptic manufacturing), FDA 21 CFR Part 211, ICH Q7 and current cGMP requirements. USP Class VI, USP87 and USP88 compliant. Batch-related certificate of conformity for each delivery.
Suitable for all cleanroom classes
Triple packaging for cleanroom classes A, B, C and D according to EU GMP Annex 1, as well as ISO 14644-1 classes 3-8. Also suitable for isolators and RABS (Restricted Access Barrier Systems). No time-consuming decontamination required for insertion.
Sterility & Certificates
Gamma-irradiated (SAL 10-⁶), sterile, DNase-free, RNase-free, pyrogen-free, endotoxin-free. USP Class VI, USP87 and USP88. Particle content tested according to USP , EP 2.9.19. Batch certificate with LOT number and expiry date on inner bag.
Customised configuration
Customised solutions on request: individual volumes (125 ml to 2,000 ml), closure options (standard, 2-port, 3-port), private labelling, own lot numbering, special packaging units according to your GMP requirements.
Technical Specifications
Volume
1,000 ml (others on request: 125-2,000 ml)
Material
PETG (polyethylene terephthalate glycol), USP Class VI
Packaging
3-fold in staggered tear-off bags (triple bagged)
Bag numbering
3 = outside, 2 = centre, 1 = inside (status display)
Sterilisation
Gamma radiation, SAL 10⁻⁶
Temperature resistance
Up to -70°C (deep-freeze storage)
Certificates
USP Class VI, USP87, USP88, DNase-free, RNase-free, pyrogen-free
Particle content
Tested according to USP , EP 2.9.19, JP 14th ed.
GMP conformity
EU GMP Annex 1, FDA 21 CFR Part 211, ICH Q7
Cleanroom classes
Class A, B, C, D (EU GMP) / ISO 3-8
Sealing
Standard, 2-port or 3-port (on request)
Traceability
LOT number and EXP date on inner bag
Applications
Wherever sterile transfer to cleanroom areas is essential.
- Aseptic biopharmaceutical production (EU GMP Annex 1)
- Vaccine & antibody production in the clean room
- Cell & Gene Therapy (CGT) Production
- Insulator and RABS applications
- Sterile filling active pharmaceutical ingredients
- Media handling in GMP class A/B
Comparison: Standard vs. Triple Bagged
| Property | Standard packaging | Triple Bagged (3-B) |
|---|---|---|
| Cleanroom class A/B | Wiping necessary | Can be used directly |
| Contamination risk | High (outer bag) | Minimal (3 barriers) |
| Time required for infiltration | High (decon process) | Very slight |
| GMP Annex 1 compliant | Conditional | Complete |
| Status display | None | Numbered bags |
| Suitable for insulators | No | Yes |

Customised GMP solutions made to measure
The innoME GmbH develops customised triple-packed consumables for your specific GMP requirements.
From the individual volume selection via special Closure concepts up to Private branding and customised lot numbering - We deliver your GMP consumables exactly as you need them.
As OEM manufacturer we take care of the entire process: development, validation, cGMP production and quality documentation - all from a single source.
Customised GMP packaging solutions
The innoME GmbH develops and produces customised triple-packaged consumables for GMP-regulated biopharmaceutical and CGT applications.
- Individual volumes (125 ml to 2,000 ml)
- PETG, PET or PC according to process requirements
- Standard, 2-port, 3-port Closures
- Private labelling with your own branding
- Customised lot numbering
- Complete GMP documentation & Batch record
- Validation support (IQ/OQ/PQ)
- Prototype to series production
How we work - your GMP process
From the enquiry to the validated series delivery - everything from a single source:
Definition of your GMP requirements: Volume, material, closure, cleanroom class, packaging units, documentation requirements
Material selection (PETG/PET/PC), validation requirements, GMP documentation, batch protocol design
Detailed offer incl. specification, certificate list, delivery times and price structure
Provision of samples for qualification in your process (IQ/OQ support)
Production under cGMP conditions with complete batch protocol, COA and certificates
Delivery with batch-related certificate of conformity, LOT number, EXP date - direct from German warehouse
Advantages
3-fold packaging - no wiping with alcohol required
EU GMP Annex 1 & FDA 21 CFR Part 211 compliant
Numbered bags - seamless status display
USP Class VI, USP87/88 - highest purity
Customised configuration - OEM & private label
German warehouse - fast delivery, no import fees
FAQ
With simply packaged products, contamination may adhere to the outside of the bag - this is brought into the cleanroom when the product is introduced and must be removed by laborious wiping with 70% alcohol. Triple-packed products eliminate this risk: each bag is opened in the corresponding cleanroom airlock, contamination remains outside. No wiping, no loss of time, no risk of contamination.
Our triple bagged products are suitable for all EU GMP cleanroom classes A, B, C and D suitable as well as for Isolators and RABS (Restricted Access Barrier Systems). The 3-fold packaging meets the requirements of the EU GMP Annex 1 (2022) for aseptic manufacturing processes.
Step 1: Outer bag 3 in the outer airlock and remove. Step 2: Middle bag 2 in the intermediate airlock and remove. Step 3: Inner bag 1 only open in the cleanroom (class A/B) - the product is guaranteed to be contamination-free. The staggered bag sizes prevent the inner bag from being damaged when the outer bag is opened.
Yes - innoME offers Triple packaging as an OEM service for various laboratory articles: PETG media bottles (125 ml to 2,000 ml), cell culture bottles, Erlenmeyer flasks and other consumables. Contact us for a customised solution according to your GMP requirements.
Each delivery contains a Batch-related certificate of conformity (COA) with LOT number, EXP date and all relevant test parameters. We can also provide this information on request: DMF (Drug Master File) information, TSE/BSE declarations, Material Safety Data Sheets, Extractables & Leachables data and validation support for IQ/OQ/PQ.